

Affiliations
ABSTRACT
Uretero-iliac artery fistula (UIAF) is a rare condition which offers a significant diagnostic challenge due to its overlapping clinical picture with other renal and urinary pathologies. We report a case of a 72-year male who presented with gross haematuria, fever and right-sided flank pain. He was initially treated on the line of pyelonephritis. The haematuria remained unsettled. An exploratory surgery was performed to look for the cause of haematuria, which detected a right-sided duodeno-renal fistula (DRF), and right-sided nephrectomy was performed. The patient’s haematuria continued after the surgery, hence a computerised tomography angiogram (CTA) was ordered which showed aneurysm of the right common iliac artery and UIAF between the right ureter and right common iliac artery. Aneurysmal resection and femoro-femoral bypass were carried out with placement of a synthetic graft.
To our best knowledge, this is the first case to be documented in English scientific literature with concomitant DRF and UIAF.
Key Words: Aneurysm, Fistula, Iliac artery, Ureter, Vascular, Concomitant.
INTRODUCTION
In 40% cases of abdominal aortic aneurysms (AAAs), iliac artery aneurysms (IAAs) are also found concomitantly.1 Isolated IAAs are a rare entity and occur at a rate of 0.4-1.9% of AAAs.1 Uretero-iliac artery fistula (UIAF) is a communication between the ureter and an iliac artery. It is a rare condition first described in 1908 by Mosch-Cowitz. Around 150 cases of UIAFs had been reported until 2015.2 The aetiology remains unclear but is mostly degenerative or atherosclerotic in 90% cases.1,3 They are classified into primary and secondary type on the basis of their etio-pathogenesis.2 Primary fistulas account for 15% of cases occurring with aorto-iliac aneurysms, vascular malformation or aberrant vessel that eroded into the ureter, while 85% cases are secondary to procedures like pelvic surgery (89%), ureteral stenting (67%) or vascular surgery.4 These interventions can lead to inflammation and fibroses that may cause fixation of the ureter to the artery. In particular, the ureteral stent can cause abrasion of the wall of ureter and radiation can causes ischaemic injury to the arterial wall by damaging the vasa vasorum.2 Other causes include malignancy, infection or connective tissue diseases.1
The main presenting symptoms are haematuria, flank pain, or both. Spontaneous in onset, massive haematuria can lead to life-threatening haemorrhagic shock in this condition. The diagnosis is often delayed because of lack of awareness of this disease and in case of internal IAA, the deep location of the artery.1,2 Open surgical and endovascular approaches are used to manage UIAF. The management involves repair of both artery and ureter involved. The optimal approach is decided depending upon the factors, like haemodynamic stability of the patient, associated co-morbidities and operative risks.4 Long-term, postoperative complications of UIAF include ischaemia (67% in surgical repair and 50% in endovascular repair), infection, stent thrombosis, deep venous thrombosis, recurrence, skin ulceration, and chronic leg pain. Antibiotic prophylaxis is recommended, although its role is uncertain.2,4 The mortality rate for UIAFs ranges from 10% to 13% and haemodynamic instability causes mortality in 22% cases.4 In case of common IAA, rupture can cause mortality in 70 % of cases. The risk of rupture increases with an increase in size, especially when it exceeds 5 cm.3 Herein, we present a rare case of a 72-year male with concomitant duodeno-renal fistula (DRF) and UIAF.
CASE REPORT
A 72-year male, with Diabetes mellitus, hypertension, and chronic obstructive pulmonary disease, was referred from another local hospital due to lack of facilities of vascular surgery. This case profile is described in two stages, starting with what the patient had gone through in his first hospital stay.
The patient complained of right lumbar pain for the past two weeks and was managed conservatively on the provisional diagnosis of right-sided pyelonephritis at a local hospital. He followed up on Day 15 at the same hospital with unremitting haematuria. His latest laboratory tests showed a hemoglobin (Hb) of 8.7 g/dL, a neutrophil leukocytosis of 20.16 ×103/uL, and platelets within physiological range. Ultrasound of abdomen and pelvis showed "particulate debris in the collecting system, with diffuse hyperechoic renal parenchyma suggestion of haemorrhage" for right kidney. Computed tomography (CT) for preceding pathology was not performed owing to his haemodynamic instability and urgency of the patient. After a comprehensive discussion between the urologists and general surgeons, the urologists decided to perform right nephrectomy via right-sided retroperitoneal flank incision. An intraoperative right-sided necrotic kidney and incidental, 4 cm long DRF was identified in the second part of the duodenum. The urologists and general surgeons worked together and proceeded with right nephrectomy, DRF repair and Roux-en-Y gastro-jejunostomy with pyloric exclusion. The DRF was between renal parenchyma and duodenum, and there was no vascular involvement. On-table cystoscopy revealed active bleeding from the right ureteric orifice. Around 700 ml of fresh and clotted blood was evacuated from the urinary bladder via cystoscopy. There were no significant intra-operative complications. The patient recovered smoothly. He was transfused two pints of whole blood, but his Hb did not improve significantly after the surgery. On the second post-operative day, he developed two episodes of hypovolemic shock with blood pressures of 80/60 mmHg and 70/60 mmHg on two different occasions with massive haematuria. The patient was resuscitated and managed on conservative lines successfully. The complete blood count (CBC) performed on the day of haematuria showed Hb of 7.5 g/dL, a white blood cell (WBC) count of 12.45×103/uL, and platelets within the normal range. A CTA was ordered on the suspicion of vascular pathology which demonstrated a 10×5 cm, fusiform aneurysm of the right common iliac artery (CIA) communicating with the right-sided ureter, and the patient was referred for vascular surgery (Figure 1).
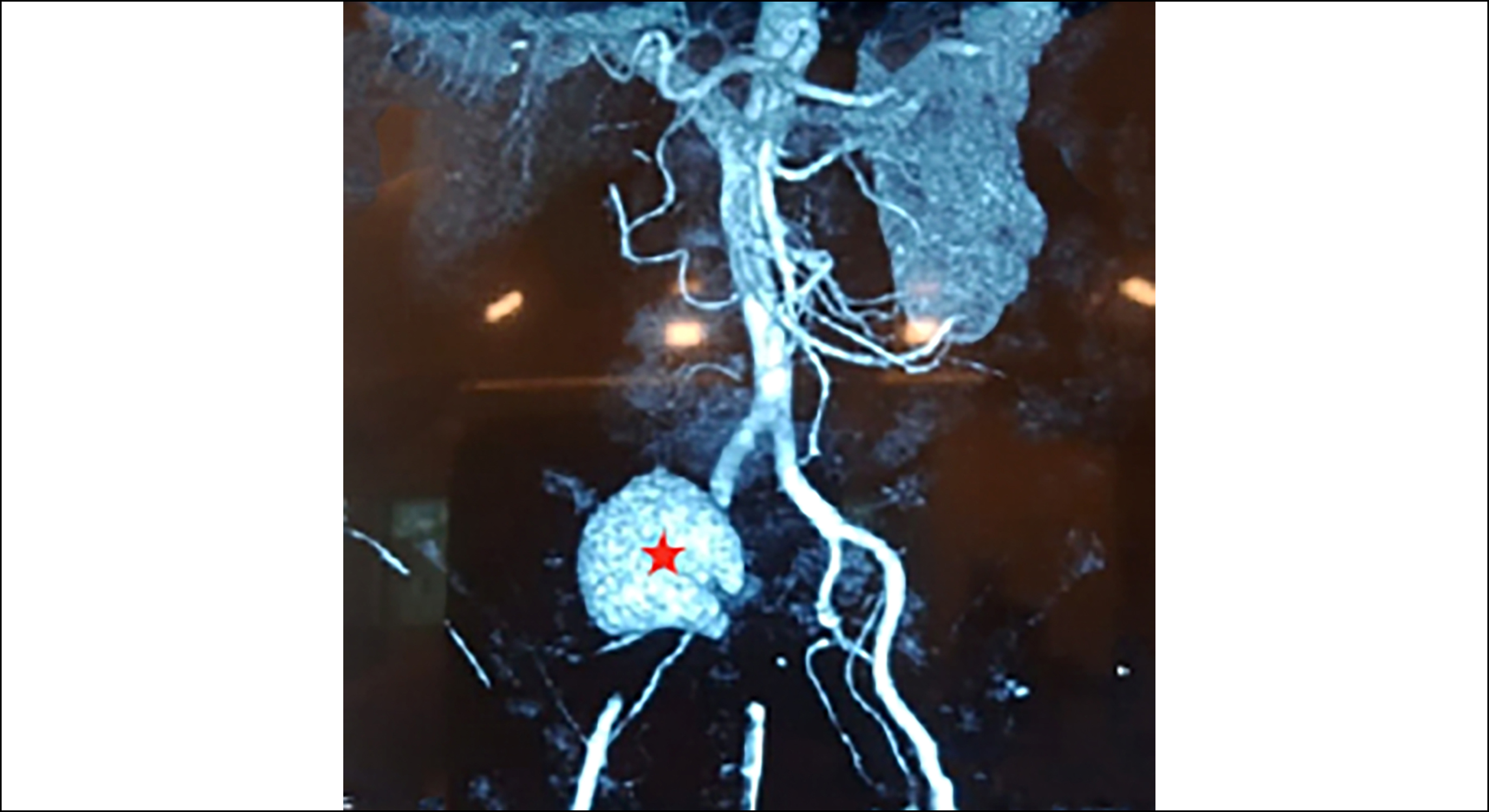 Figure 1. Computerised tomography angiogram (CTA) showing right common iliac artery aneurysm (red star) in sagittal section.
Figure 1. Computerised tomography angiogram (CTA) showing right common iliac artery aneurysm (red star) in sagittal section.
The receiving surgeons optimised the patient and decided to explore and repair the aforementioned aneurysm, suspecting it as the cause of massive haematuria and hypovolemic shock. The surgeons preferred to explore the patient through previous incision. An operative repair of the right uretero-iliac fistula (UIF) was done, and proximal right CIA, external iliac artery (EIA) and internal iliac artery (IIA) were ligated, and the aneurysmal cavity closed with Vicryl 2-0. A 22 French drain was placed into the cavity. Bilateral groin was explored to carry out femoro-femoral crossover with expanded polytetraflouroethylene (ePTFE) graft to restore blood supply of the right leg (Figure 2). There were no intraoperative complications. The patient had a smooth recovery. There were no immediate postoperative complications. The patient was well-managed during his ward stay. He had no subsequent episodes of haematuria, his bilateral distal lower limb pulses were palpable and the wounds were healing well. He was discharged to home on 10th postoperative day.
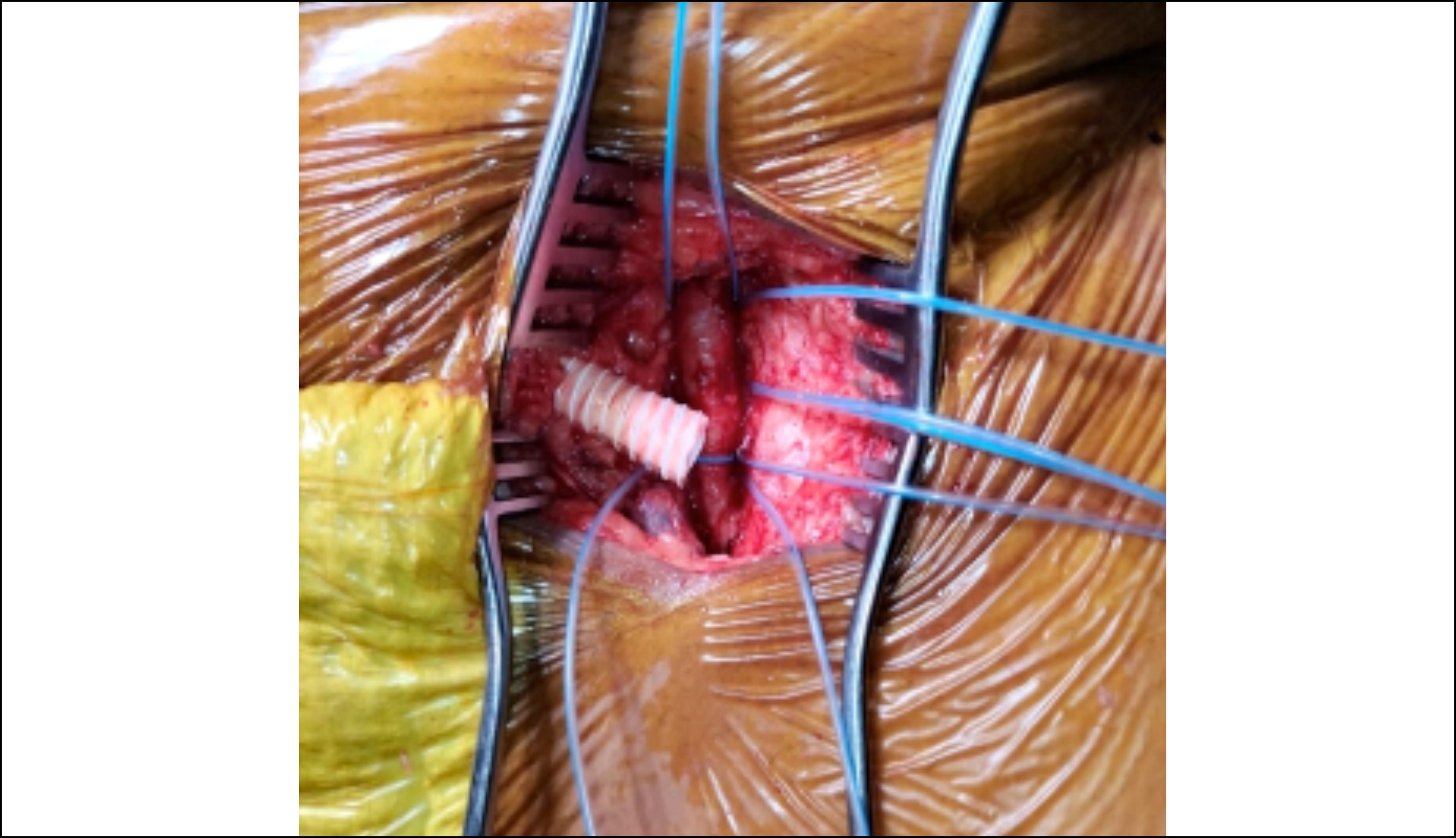 Figure 2: Placement of expanded polytetraflouroethylene (ePTFE) graft before femoro-femoral cross-over.
Figure 2: Placement of expanded polytetraflouroethylene (ePTFE) graft before femoro-femoral cross-over.
The biopsy of aneurysmal wall showed non-specific necrosis, and no microbial growth was appreciated on culture and sensitivity testing. The blood culture showed no growth after 7 days of incubation at optimal temperature and pressure settings. Lately, his kidney biopsy revealed an acutely inflamed, necrotic kidney, containing septate fungal hyphae suggestive of Aspergillus fumigatus. He was prescribed anti-fungal drugs and has been treated accordingly by urologists.
Till date, he has developed no postoperative neurovascular deficits in his limbs and no episodes of haematuria. His post-vascular surgery sequelae has been monitored in the follow-up. His wounds have healed well. He was being followed up weekly for one month, fortnightly for another month, and will be followed on monthly basis till six months. During two of his visits, we found normal CBCs of the patient.
DISCUSSION
The risk factors of male gender, advanced age, hypertension and smoking, held causative in the pathogenesis of large vessel aneurysms were present in this patient with CIA aneurysm (CIAA).5 Mrad et al. documented similar findings of majority of male patients afflicted with this type of aneurysm.3 However, a review article by Pillai et al. demonstrated female preponderance.2 The age of presentation of this patient was 72 years, similar to the mean age of presentation of 72 years described by Huang et al. and different from the study by Pillai et al., i.e. 61.3 years.2,6
The most frequently documented clinical features at presentation of CIAAs with or without complications are lower abdominal pain, haematuria, and flank pain.2,6 About 70% of patients present with the clinical sign of pulsatile mass in abdomen.5 However, the absence of this finding in the current patient cannot rule out the condition.
Secondary cases of UIAF are attributed to malignancies, most commonly to gynaecologic cancers (28%) and chronic ureteral stents (73.7%).2,4 The primary variety of CIAA has association with simultaneous aortic aneurysm, aberrant vessel aneurysm or isolated CIAA eroding the ureter, the latter being the finding in this case.2 Some studies show the phenomenon of “ureteral encasement” by CIAA, whereby the ureter gets embedded in the aneurysm. The encasement can cause urinary tract infections (UTIs), bacteremia, septicemia, and haematuria.7 Distal CIA is the most commonly reported site of aneurysm.2 The annual expansion rate of CIAAs is found to be 2.9 mm/year, with hypertension accelerating the rate of expansion.6
An early diagnosis is paramount to lower the mortality risk. CTA offers immediate treatment strategy and helps in locating the lesion and defining the size of the aneurysm.1,4,5 Pillai et al. document digital subtraction angiography (DSA) to be the best imaging modality for CIAAs.2 The previous studies reported high mortality rate (64%) in patients who underwent exploratory laparotomy without prior diagnosis. Hence, the initial work-up should include modalities to enable a prompt diagnosis. These include diagnostic ureteroscopy, retrograde ureteropyelography, CTA, and ultrasonography.8
Operative intervention for CIAAs is indicated in two studies at a cut-off diameter of 30 mm.3,6 The symptomatic cases are also operated, as was the case of unremitting haematuria in this elderly patient. A multi-disciplinary team, involving urologists, vascular surgeons, and radiologists is highly advocated in UIAFs.8 Two approaches are opted to manage CIAAs with or without complications. Open repair techniques include primary repair, ligation of the artery and suturing with a graft and urinary diversion, nephrostomy with bypass and trans-renal ureteral occlusion with Gianturco coils or selective vascular embolisation with extra anatomical vascular management.4 If the kidney function is inadequate, then nephrectomy can also be done.8 Recently, the endovascular management has become the treatment of choice because of its minimally invasive nature.3 Endovascular procedures have relatively low associated risks, shorter hospital stay, and the possibility of conduction of procedure under local anaesthesia.3,8
However, an American study demonstrated a successful management of 90% cases of CIAAs with open surgical repair. It also showed the superiority of open surgical repair over endovascular repair in terms of graft failure rate, i.e. 1% and 5%, respectively. In the current case, open surgical repair for CIAA was preferred due to the haemodynamic instability of the patient and concomitant UIAF.6
Stent-grafts exclude the fistula and maintain distal perfusion post-procedure. The commercially used stent-grafts include the Viabahn, iCast, Fluency, and Wall-graft. Wall-graft stent was used in this patient, made up of ePTFE.2
Huang et al. documented pulmonary complications (15%) and buttock claudication (8%) topping all the immediate and late post-procedural complications in CIAA surgeries, respectively.6 Pillai et al. reported lower limb ischaemia (57%) manifesting as ulcers, deep venous thrombosis, leg pain, and osteonecrosis as late complications following the surgical repair of CIAAs. 2
In conclusion, UAF should be included in the list of differential diagnoses of presenting cases of unremitting haematuria. A high index of suspicion is required to make an accurate diagnosis of and manage UIAF promptly. Angiography yields the highest diagnostic outcomes. Timely surgical correction done either by endovascular stenting or open surgical repair, as in this case, can prevent deleterious complications.
PATIENTS’ CONSENT:
Informed consent was taken before writing this case report and its disclosure.
COMPETING INTEREST:
The authors declared no conflict of interest.
AUTHORS’ CONTRIBUTION:
IS, MN: Substantial contributions to the conception or design of the work, reviewing the work critically for important intellectual content, final approval of the version to be published, and agreed to be accountable for all aspects of the work.
RZK, MFC: Substantial contribution to the acquisition of data for the work, drafting the work, final approval of the version to be published, and agreed to be accountable for all aspects of the work.
HMQ: Substantial contribution to the conception of the work, acquisition, analysis, and interpretation of data for the work, drafting the work and reviewing it critically for important intellectual content, of final approval of the version to be published, and agreed to be accountable for all aspects of the work.
All authors approved the final version of the manuscript to be published.
REFERENCES
Copyright © 2025. The author(s); published by College of Physicians and Surgeons Pakistan. This is an open-access article distributed under the terms of the CreativeCommons Attribution License (CC BY-NC-ND) 4.0 https://creativecommons.org/licenses/by-nc-nd/4.0/ which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.