

Affiliations
ABSTRACT
Cardiac metastases are the most common cardiac tumours. Any malignant tumour can metastasise to heart, but the most common causes of cardiac metastases are melanoma, lung, breast, oesophageal, and haematological malignancies. Endometrial stromal sarcoma (ESS) is a very rare endometrial malignancy. Metastases are usually found in the abdominal cavity, particularly on the peritoneal surfaces. Rarely, metastases to distant organs have also been reported. Cardiac metastases of ESS are extremely rare. Herein, we present a case of ESS who underwent successful surgery for right atrial metastasis 23 months after the initial diagnosis.
Key Words: Endometrial stromal sarcoma, Cardiac tumour, Cardiac metastasis.
INTRODUCTION
Cardiac metastases are 20 to 40 times more common than primary cardiac tumours.1 Cardiac tumours were found in up to 20% of autopsy series performed on patients with a known malignancy.2 Any malignant tumour can metastasize to the heart, but the leading causes of cardiac metastases are melanoma, lung, breast, oesophageal carcinomas, and haematological malignancies.3 Endometrial stromal sarcoma (ESS) is a very rare malignant tumour, accounting for approximately 0.2% of all uterine malignancies. The annual incidence of ESS is 1-2 cases per million women. It usually affects perimenopausal women but occasionally young women and adolescents.4 The site of recurrence is usually the pelvic region. Metastases are usually seen in the abdominal cavity, especially on the peritoneal surfaces. Distant organ metastases have been reported rarely. Cardiac ESS metastases are extremely rare. Herein, we present a case of ESS who underwent successful surgery for right atrial metastasis 23 months after the initial diagnosis.
CASE REPORT
A 39-year female, who was premenopausal before diagnosis, presented to the hospital because of pelvic pain in January 2020. A cystic lesion of 3 cm in the uterus and 1.3 cm in the right adnexa was detected on imaging.
Total abdominal hysterectomy and unilateral salpingo-oophorectomy were performed. ESS was confirmed by immunohistochemistry (IHC) on histopathological examination. Tumour cells were positive for estrogen receptor (ER), progesterone receptor (PR), and CD10. She did not receive any adjuvant treatment and was followed up. She then registered for a follow-up visit at the oncology clinic in December 2021. A well-circumscribed, homogeneous mass lesion of 4.5x4 cm filling approximately 50% of the right atrium was identified on chest CT at this visit (Figure 1).
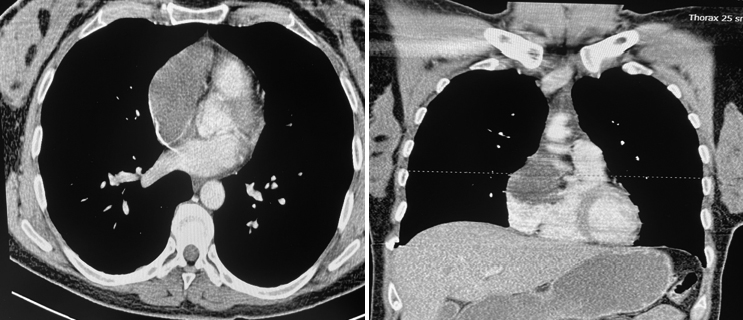 Figure 1: Chest CT shows a mass lesion of the right atrium.
Figure 1: Chest CT shows a mass lesion of the right atrium.
The PET scan performed in March 2022 showed no findings except for the appearance of a 6.0x4.0 cm hypodense hypermetabolic lesion (SUV-max 12.67) in the right atrium. The patient was operated on March 29, 2022. Open heart surgery was performed with a minimally invasive technique. The patient's heart was reached through a right anterolateral thoracotomy (8 cm incision). A patch was prepared from the patient's own pericardium. A solid, lobulated mass of 6x8 cm, growing towards the superior vena cava, localised in the right atrial appendage and partially covered by a thrombus, was identified and excised in one piece without fragmentation, together with the atrial tissue to which it was attached. The remaining atrial tissue was cauterised, and the right atrial anterior wall was reconstructed with a pericardial patch to ensure tissue and functional integrity. The surgically removed lesion was histomorphologically interpreted as "metastatic ESS". IHC staining showed that these cells were positive for CD10, cyclin D1, ER, and PR.
(a) Spindle-shaped neoplastic cells infiltrating myocardium (H&E, x400, (b) Diffuse immunoreactivity in neoplastic cells (cD10, 1HC), (c) Increased mitotic activity in neoplastic cells (H&E, x400), (d) Diffuse immunoreactivity in neoplastic cells (Cyclin D1, 1HC).
She received 4 cycles of gemcitabine-docetaxel chemotherapy. The patient, who had no metastases on imaging, is being followed with letrozole + LHRH therapy.
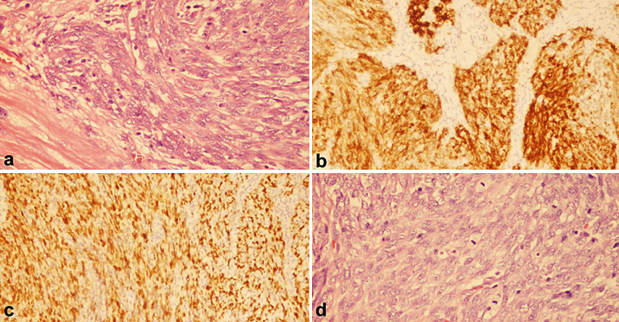 Figure 2: Histopathology of the cardiac mass.
Figure 2: Histopathology of the cardiac mass.
DISCUSSION
Metastases are the most common cardiac malignant masses.5 Since cardiac metastases develop at a very late stage in patients with malignancy, they are not frequently observed on imaging and have been found in only 12-25% of cases in autopsy series.6 Tumours can reach the heart in four ways: hematogenous spread, lymphatic spread, transvenous spread, or direct invasion. Hematogenous spread usually leads to myocardial or endocardial metastases.7 The spread of melanoma and haematological malignancies are examples of this. Masses that spread through the lymphatics often seed the pericardium or epicardium. Lymphatic spread is noted in epithelial tumours such as breast and lung cancer. Locally aggressive mediastinal and pleural tumours such as mesothelioma, can directly invade the pericardial sac. Renal cell carcinoma and uterine sarcomas may extend into the inferior vena cava and grow into the right atrium by transvenous spread.8 The presence of an intracavitary mass in the right atrium in our patient suggested a transvenous spread. Information on cardiac metastases from ESS is available in the literature in the form of rare case reports.9 In the presented case, a cardiac mass was detected on the chest CT of the patient, who had previously been operated on for ESS. The case is interesting in such a way, that the patient was asymptomatic and had isolated cardiac metastases. If a mass is detected in the heart of patients with known malignancies, metastasis to the heart should be considered first.
PATIENT'S CONSENT:
Written informed consent was obtained from the patient.
COMPETING INTEREST:
The authors declared no competing interest.
AUTHORS’ CONTRIBUTION:
ST, DSK, VB, SR, OY: All the authors contributed to the design, drafting, and critical revision of the manuscript.
All the authors have approved the final version of the manuscript to be published.
REFERENCES
Copyright © 2025. The author(s); published by College of Physicians and Surgeons Pakistan. This is an open-access article distributed under the terms of the CreativeCommons Attribution License (CC BY-NC-ND) 4.0 https://creativecommons.org/licenses/by-nc-nd/4.0/ which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.